ReadyMedical
EFFICACY TESTS

REVITALIZING: HA+GF
Human fibroblasts have been treated in vitro with ReadyMedical special compound of sonicated Hyaluronic Acid + Peptides with the wound healing technique. After 24 hours the treated cells had increased their motility and proliferation of up to 44%, compared to the untreated cells used as negative controls (1).
Using the inserts, a gap of 500 μm was created in a confluent cell colture monolayer and the wound closure was evaluated by measuring the width of injury line, taking pictures at regular interval of 24 hours.
The effect of the Product ReadyMedical HA + GF was evaluated as the capability of cells to reduce the original gap of 500 μm (100% of wound closure), comparing the pictures, taken at different times with the corresponding pictures of the initial wounded monolayer. The result has been the complete wound closure of the cells treated with ReadyMedical HA + GF after 72 hours.
1) UB-CARE – UNIVERSITY OF PAVIA. Report nr. PRL/17/LC1/P2. Date: 25/07/2017. The full version of this study is available upon request.
POST-TREATMENT: LENITIVE HA+PFD+PA
The study has been conducted through the in vitro evaluation of the anti-inflammatory activity of ReadyMedical HA+PFD+PA (1) on human fibroblasts.
A human keratinocyte cell line has been treated with this special compound of sonicated Hyaluronic Acid + Perfluorodecalin + Physalis Angulata. The treated cells showed a decrease of ~ 94% of inflammation compared to the untreated cells used as negative control.
The anti-inflammatory activity has been calculated measuring the IL-8 levels 24 hours after treating the fibroblasts with the product; the result was a decrease of the IL-8 levels between 93,25% and 94,82%, compared to the untreated cells.
IL-8 is the most direct and reliable marker of inflammation among the wide cytokine family. Cytokines are small proteins known to play an important role in the immune system; in particular they are able to influence the activation, maturation, growth and differentiation of several cell populations. IL-8 secretion is particularly active in chronic inflammation conditions, thus measurement of this cytokine in an in vitro model, composed by fibroblasts, represents a useful and sensitive tool to assess the anti-inflammatory effect of a topical product.
1) UB-CARE – UNIVERSITY OF PAVIA. Report nr. AIF/17/LC1/P1. Date: 12/07/2017. The full version of this study is available upon request.
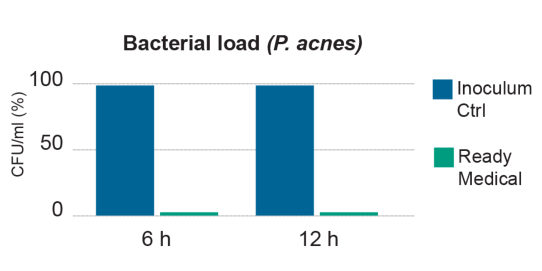
ACNYCID: HA + AC
WITH READYMEDICAL ACNYCID HA + AC